Advancing lactase deficiency through genetic testing
Eralydix develops an innovative genetic testing technology designed to improve lactase deficiency by identifying its genetic cause.
By combining advanced DNA analysis with a simplified testing process, our solution enables more precise diagnosis and supports better medical decision-making.
The scientific foundation of lactase deficiency
Lactose intolerance is primarily caused by a reduction in lactase enzyme activity, often linked to genetic variations.
Understanding these biological mechanisms is essential for advancing lactase deficiency and improving patient care.
Eralydix focuses on detecting these genetic variations directly, providing a reliable and science-based diagnostic approach.
A new approach to lactase deficiency
Our core innovation lies in targeting the genetic causes of lactose intolerance rather than relying on indirect diagnostic methods.
This allows for a more precise identification of the pathology related to lactase deficiency and promotes a more personalized approach.
🎯 Precision
⚡ Speed
🧬 Genetic insight
How our technology supports works
Our assay detects specific genetic markers associated with lactase deficiency through a streamlined DNA analysis process.
This enables accurate diagnosis without invasive procedures and provides reliable data to guide lactase deficiency strategies.
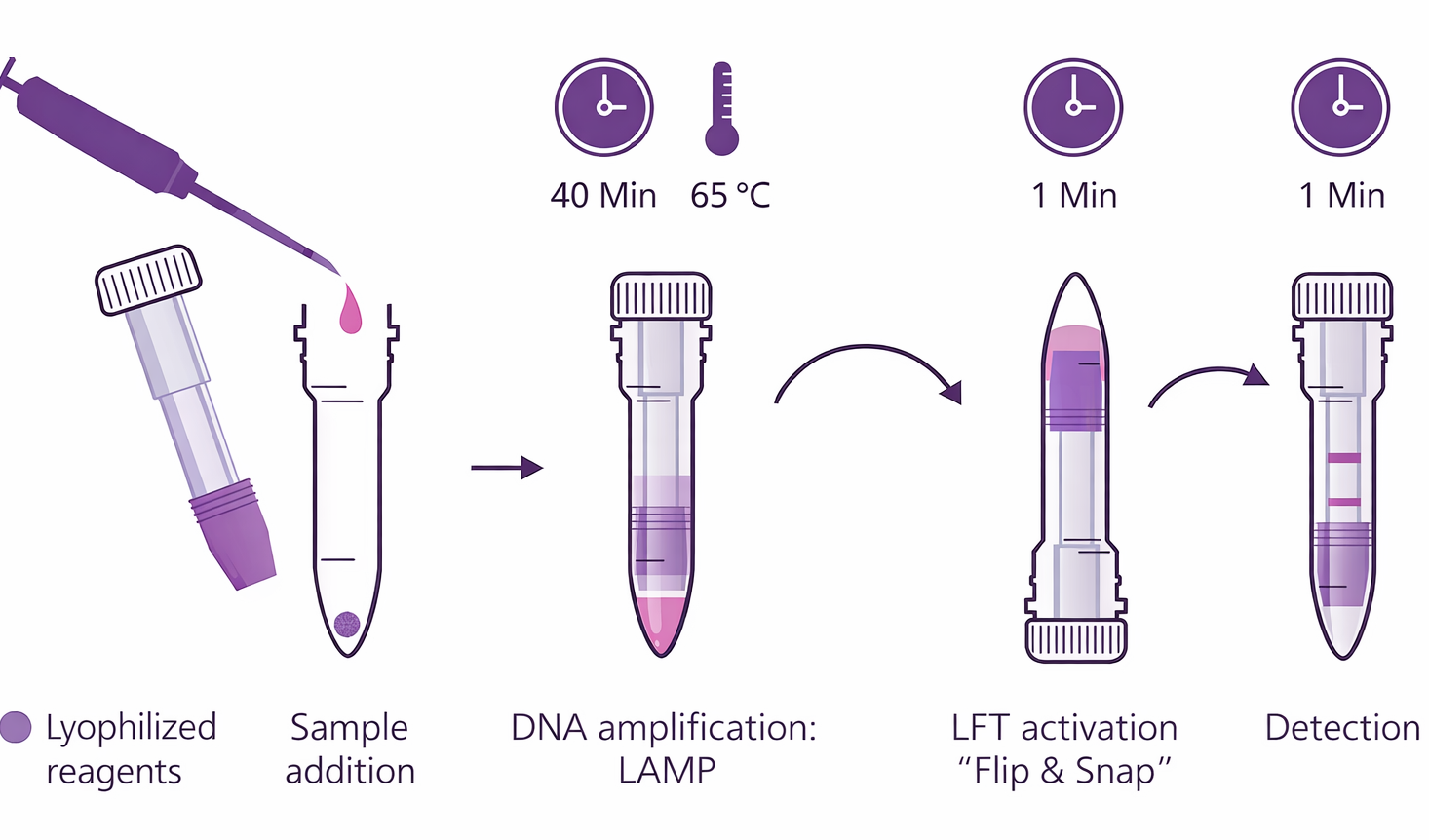
🧫 Lamp Amplification
Isothermal Loop-Mediated Amplification for rapid DNA processing targeting C/T-13910 and G/A-22018 genes
🧬 LFA Visual Detection
Uses Lateral FlowAssay Technology to provide clear and easy-to-read screening results
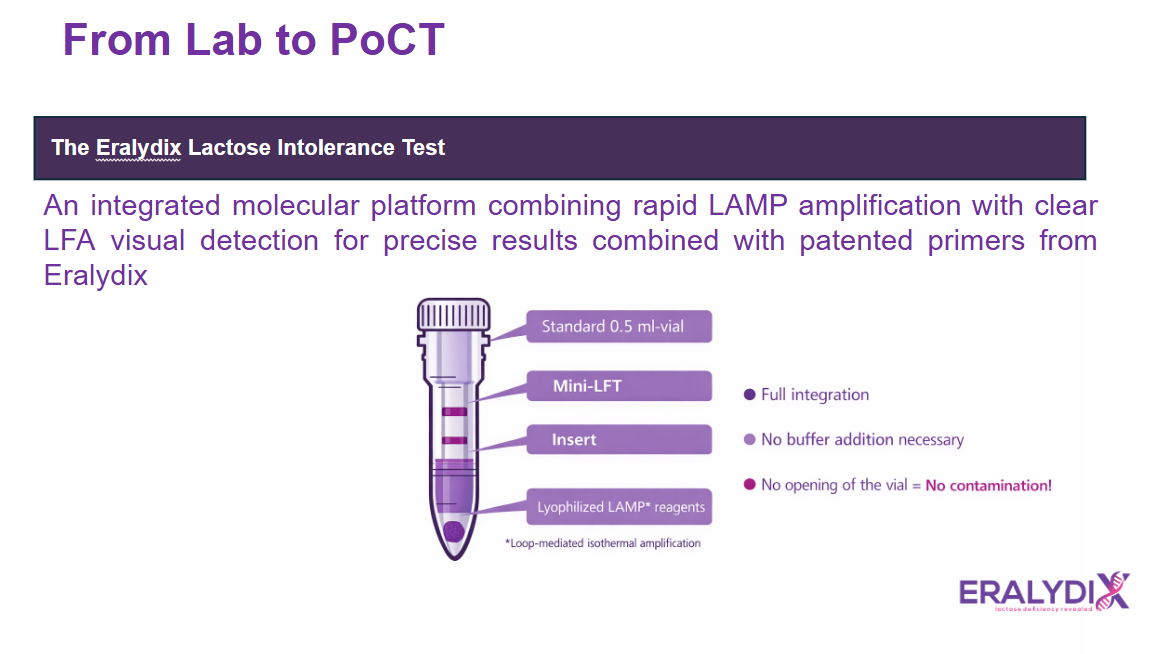
📊 Eralydix CampyTube Platform
A closed integrated system that minimizes contamination and streamlines the testing workflow
Technical advantages of our product
Our solution has been designed to overcome the limitations of traditional diagnostic methods by making genetic testing more accessible, efficient and scalable.
By reducing infrastructure requirements and simplifying workflows, Eralydix enables broader adoption across healthcare systems while opening new opportunities in both developed and emerging markets
⚡ Fast and reliable results
🏥 Potential for large-scale deployment
✔️ Simple and efficient workflow
🔬 Robust and scientifically grounded approach
A better alternative to traditional diagnostic methods
Current diagnostic approaches include breath tests and empirical dietary trials, which can be unreliable or uncomfortable.
Eralydix differentiates itself by:
targeting the genetic origin of lactose intolerance
providing more accurate and consistent results
enabling personalized lactose intolerance treatment
| Hydrogen H2 Breath test | Lactose tolerance test | Eralydix LAMP LactoCheck | Duodenal Lactase activity | Serum or urine gaxilose/galactose test | Genetic test | |
|---|---|---|---|---|---|---|
| Lactose challenge | YES | YES | NO | NO | YES | NO |
| Assessment of Symptoms | YES | YES | NO | YES | NO | NO |
| Test Principle | Detection of H2 in expiratory after lactose challenge | Increase of in plasma glucose after lactose challenge | Detection of 13910(C/T) polymorphism in Portable/Home Rapid Test | Lactose enzymatic activity in duodenal biopsy | Detection of D-xylose or Galactose (blood or urine) after lactose challenge | Detection of 13910(C/T) polymorphism in Laboratory PCR testing |
| Cost | ++ | + | + | ++++ | +++ | +++ |
| Skilled staff & Equipment | +++ | ++ | + | ++++ | ++ | +++ |
| Limitations | False negative tests by H2 non producers; False positive test in rapid transit or altered bowel anatomy | Glucose disorders and altered bowel anatomy | False negative in presence of atypical mutations | Patchy expression of lactase; endoscopy shouldn't be performed as a primary intention | Variable test performance in the literature. False positive in various conditions such as rapid transit and others | False negative in presence of atypical mutations |
| Invasive & side effect | Low invasive but strong and side effects | Low invasive (blood sampling) but strong and side effects | Non invasive (buccal swap) no side effect | Highly invasive and potential side effect due to sedation or anaesthesia | Low invasive (blood sampling) High side effects | Low invasive (depend on sampling) no side effect |
| Sensitivity (literature avg) | 77–88% | 75–94% | >90% expected | 95–100% | 70–90% | 90–100% |
| Specificity (literature avg) | 85–98% | 77–96% | >90% expected | 95–100% | 80–95% | 95–100% |
| Cost estimation in CH | CHF 400.- incl med. visit | CHF 250.- incl med. visit | CHF 50-100.- | CHF 2'000.- | Only research purpose | CHF 400.- incl med. visit |
Positioning in next-generation lactase deficiency
Eralydix positions itself as a key player in the discovery of lactase deficiency by integrating genetic knowledge into diagnostic processes.
Our technology bridges the gap between scientific research and practical clinical application, enabling more accurate and personalized care.
Let’s explore the genetic identification of lactase deficiency.
Our Eralydix’s technology FAQ
🧬 How does Eralydix’s technology work?
Eralydix’s technology analyzes genetic markers associated with lactase deficiency to provide clear and actionable diagnostic insights. The process is designed to be simple, fast and reliable.
🧪 What makes this technology innovative?
Our technology removes the need for complex lab infrastructure and enables faster, more accessible genetic testing. It represents a significant advancement in biotech innovation applied to diagnostic medicine.
⚡ Is this technology scalable?
Yes, Eralydix’s technology is designed to be scalable across different healthcare environments, from laboratories to point-of-care settings, making it adaptable to global markets.
🩺 What are the main advantages of this technology?
The main advantages include speed, accessibility, cost-efficiency and the ability to deliver precise genetic insights, making it a strong alternative to traditional diagnostic methods.